What Is Monovisc?
Monovisc® is a single-injection hyaluronic acid (HA) viscosupplement designed to relieve joint pain caused by osteoarthritis (OA). It is composed of a high molecular weight, ultra-pure sodium hyaluronate derived from a non-animal source and cross-linked for enhanced durability.
Monovisc is indicated for the treatment of mild to moderate osteoarthritis symptoms of the shoulder, hip, knee, and ankle joints in the European Economic area and Canada, and FDA-approved for use in the knee. It offers long-lasting relief with just one injection.1
Real Life Advantages
Clinically Proven Long-Lasting Pain Relief
- Long-lasting pain relief: HA provides long-lasting pain relief greater than 6 months5
- Proven patient outcomes: Significant improvements in pain and physical function4
- Safe and effective: Strong safety profile in both an initial and repeat injection1
- Proven safety profile: No device related serious adverse events4
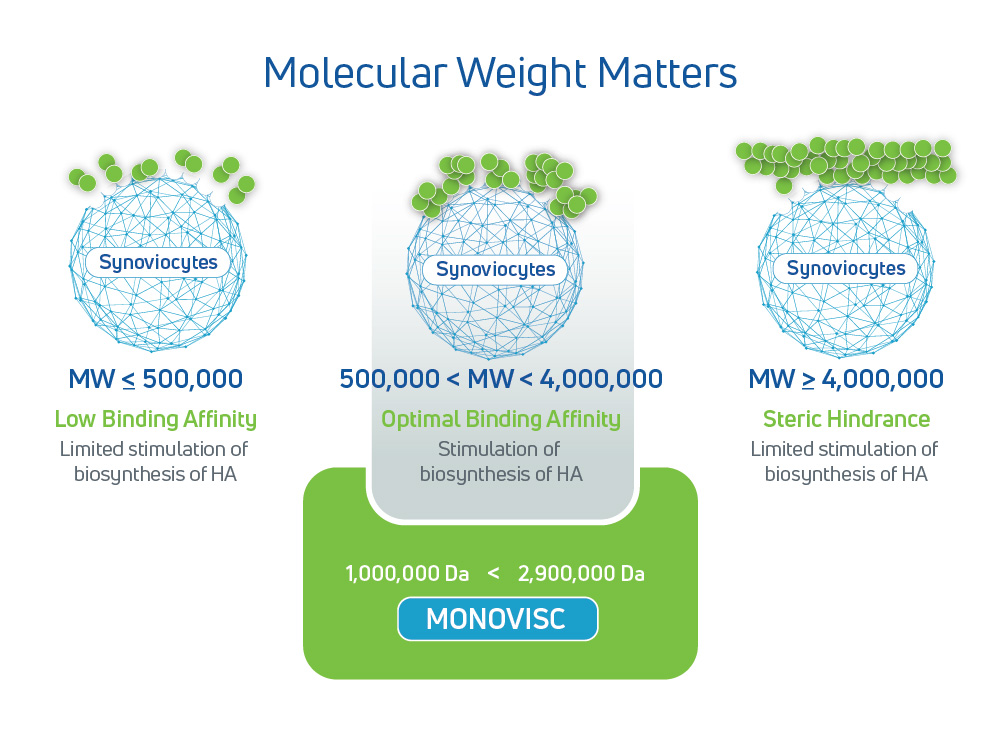
Advanced Hyaluronic Acid (HA) Formulation
- Natural solution: Highly concentrated, non-animal-based HA produced from bacterial fermentation1
- High HA concentration: 22 mg/mL, one of the highest available in its class
- Proprietary HA formulation: Lightly cross-linked to increase residence time1
- Biocompatible and resorbable: Safely and naturally broken down by the body
- Extended residence time: Patented cross-linking slows down degradation to stay in the joint longer so its actions last longer, leading to longer paint relief
Convenience & Availability
- Convenient single-injection treatment: Simple injection administered during a routine office visit with no need for multiple visits
- Global availability: Registered and approved in over 45 countries worldwide

Evidence
Monovisc received FDA approval based on a pivotal, randomized, double-blind, controlled clinical trial (Protocol 0702) involving 369 patients across 31 centers in the U.S. and Canada. The study demonstrated statistically significant improvements in pain relief and joint function compared to saline placebo over a 26-week period.4
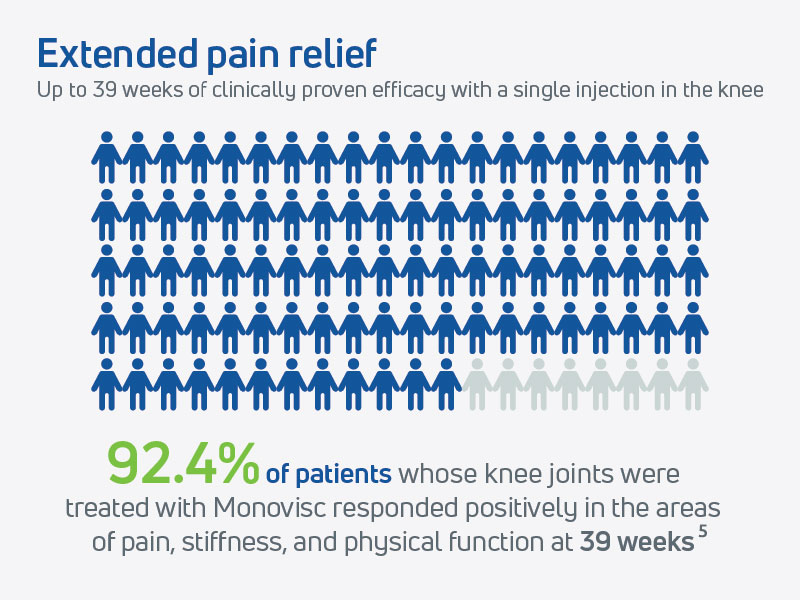
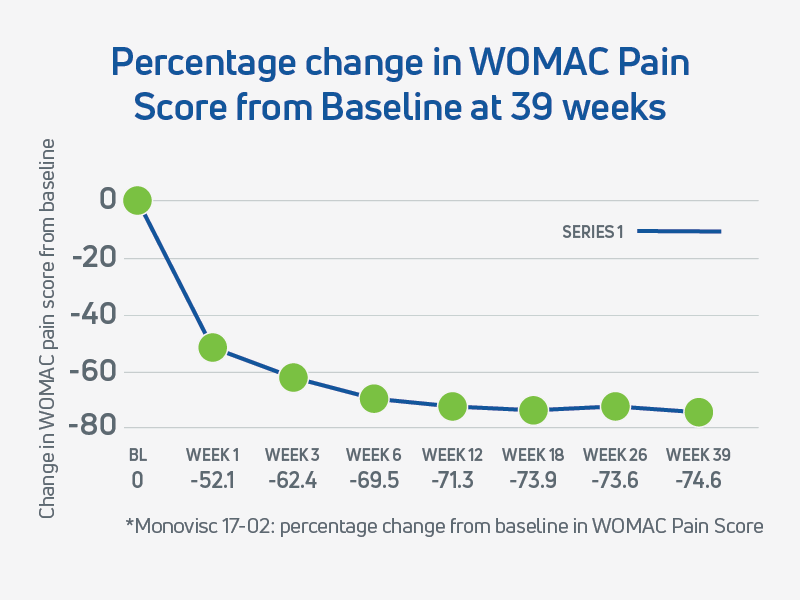
Further supporting its long-term effectiveness, a post-hoc analysis (Study 17-02) revealed that a single injection of Monovisc provided extended pain relief lasting up to 39 weeks in a substantial subset of patients.5 The extended duration of pain relief and other benefits further highlights Monovisc’s potential to delay the need for more invasive interventions.
In addition, real-world post-approval studies have confirmed Monovisc’s favorable safety profile and consistent clinical performance in broader patient populations, reinforcing its role as a reliable, non-surgical treatment option for knee osteoarthritis.1
Across the shoulder, hip, ankle, and knee, clinical studies have demonstrated a minimum 80% OMERACT-OARSI responder rate at 6 months, underscoring Monovisc’s effectiveness in multiple joints.6-9
Since its launch, about 5 million Monovisc injections have been performed worldwide.10 This reflects its strong clinical adoption and effectiveness in providing long-lasting pain relief and improved joint function for patients with osteoarthritis.10


Select Publications & Clinical Data
Stephanie C. Petterson, Kevin D. Plancher
Key Takeaways
- Multicenter, double-blind, randomized, placebo-controlled trial evaluating a single intra-articular injection of lightly cross-linked hyaluronic acid (Monovisc) in patients with symptomatic knee osteoarthritis.
- 369 patients randomized to receive either Monovisc or saline; outcomes assessed over 26 weeks using WOMAC pain and function scores.
- Monovisc group showed significantly greater pain reduction (≥50% improvement and ≥20 mm absolute improvement) compared to placebo (p = 0.043).
- Clinically meaningful pain relief observed within 2 weeks, with sustained benefits through 26 weeks.
- Safe and well-tolerated, with minimal adverse events and high patient compliance due to single-injection convenience.
- Conclusion: Monovisc is a safe, effective, and convenient treatment option for knee OA, offering rapid and sustained symptom relief.
J&J Medtech
Key Takeaways
- Review of pivotal clinical trials and post-marketing safety data comparing FDA-approved single-injection hyaluronic acid (HA) products for knee osteoarthritis.
- Monovisc contains high molecular weight, cross-linked HA delivered in a single 4 mL injection (88 mg), offering high concentration and volume.
- Demonstrated clinically significant pain relief and functional improvement in OA patients, with effects lasting up to 26 weeks.
- Favorable safety profile with low incidence of adverse events across studies and post-marketing surveillance.
- Compared to other single-injection HA products, Monovisc may offer an optimal balance of efficacy, safety, and convenience for many patients.
- Conclusion: Monovisc is a well-tolerated, effective treatment option for knee OA, with strong clinical and safety data supporting its use.
Cingal17-02
Key Takeaways
- The WOMAC pain score remained improved, and statistically significant from baseline through to 39 weeks in the Monovisc group, supporting the durability of effect from a single injection.
Resources
Product Information
Access comprehensive technical documentation and marketing materials
- Brochure – knee
- Brochure – extended indications
Instructions for Use
Complete product specifications and safety information
Looking for Monovisc in the US?
In the United States, Monovisc and Orthovisc are exclusively distributed by J&J MedTech.

To learn more about these products or connect with a representative, please visit the J&J MedTech website(opens in a new tab).
In the U.S., Monovisc is indicated for the treatment of pain in osteoarthritis (OA) of the knee in patients who have failed to respond adequately to conservative non-pharmacologic therapy and to simple analgesics, e.g. acetaminophen.12
In Canada, Monovisc is indicated in the treatment of pain in osteoarthritis (OA) of the knee, hip, shoulder, and ankle for patients who have failed to respond adequately to conservative non-pharmacologic therapy and to simple analgesics, e.g. acetaminophen.13
In the European Economic Area, Monovisc is indicated as a viscoelastic supplement or a replacement for synovial fluid in human knee, hip, shoulder, and ankle joints. Monovisc is well suited for treatment of the symptoms of mild to moderate osteoarthritis of the knee, hip, shoulder, and ankle joints for patients who have failed to respond adequately to conservative non-pharmacologic therapy and simple analgesics. In clinical trials, Monovisc has been proven to have a duration of effect of at least 6 months and a second injection 6 months after the first injection was shown to be safe.1
Anika products may not be available in all geographies. Product availability is subject to the regulatory clearances in individual markets. Please reach out to your local representative or Contact Us if you have questions about specific market approvals.
Important Safety Information
MONOVISC should not be taken if you are allergic to hyaluronate products. If you have any known allergies, you should consult with your healthcare professional. You should not have an injection into the knee if you have infections or skin diseases around the injection site. Possible side effects of MONOVISC injections in the knee joint: pain, swelling, heat, rash, itching, bruising and/or redness. You may also feel achy. These reactions are generally mild and do not last long. If any of these symptoms or signs appear after you are given MONOVISC or if you have any other problems, you should call your healthcare professional.
For complete product information, including indications, contraindications, warnings, and precautions, please refer to the Instructions for Use found here(opens in a new tab).
- AML-3000172 REV-A IFU
- Monovisc Summary Of Safety And Effectiveness Data
- Garantziotis S, Savani RC. Hyaluronan biology: A complex balancing act of structure, function, location and context. Matrix Biol. 2019 May;78-79:1-10. 9 https://pmc.ncbi.nlm.nih.gov/articles/PMC6774756/pdf/nihms-1540392.pdf(opens in a new tab)
- Stephanie C. Petterson, Kevin D. Plancher Single intra-articular injection of lightly crosslinked hyaluronic acid reduces knee pain in symptomatic knee osteoarthritis: a multicenter, double-blind, randomized, placebo-controlled trial; Knee Surgery, Sports Traumatology, Arthroscopy https://pubmed.ncbi.nlm.nih.gov/30159738/(opens in a new tab)
- 17-02 Data on file https://clinicaltrials.gov/study/NCT03390036?cond=Osteoarthritis&term=knee%2017-02&intr=Cingal%20&rank=1&tab=results(opens in a new tab)
- 13-01 Data on file https://clinicaltrials.gov/study/NCT01891396?cond=Osteoarthritis&term=Cingal%2013-01&intr=Cingal&rank=2&tab=results(opens in a new tab)
- 18-01 Data on file (hip) https://clinicaltrials.gov/study/NCT04204083?cond=Osteoarthritis&term=Monovisc%2018-01&intr=Monovisc&limit=10&sort=@relevance&rank=1&tab=results(opens in a new tab)
- 18-02 Data on file (shoulder) https://clinicaltrials.gov/study/NCT04204265?cond=Osteoarthritis&term=Monovisc%2018-02&intr=Monovisc&limit=10&sort=@relevance&rank=1&tab=results(opens in a new tab)
- 18-03 Data on file (ankle) https://clinicaltrials.gov/study/NCT04204278?cond=Osteoarthritis&term=Monovisc%2018-03&intr=Monovisc&limit=10&sort=@relevance&rank=1&tab=results(opens in a new tab)
- Data on file, Anika Therapeutics, Inc.
- Smith MM, Ghosh P. The synthesis of hyaluronic acid by human synovial fibroblasts is influenced by the nature of the hyaluronate in the extracellular environment. Rheumatol Int. 1987; 7(3):113-22 https://pubmed.ncbi.nlm.nih.gov/3671989/(opens in a new tab)
- AML-500-299/E IFU
- AML-500-260/C IFU